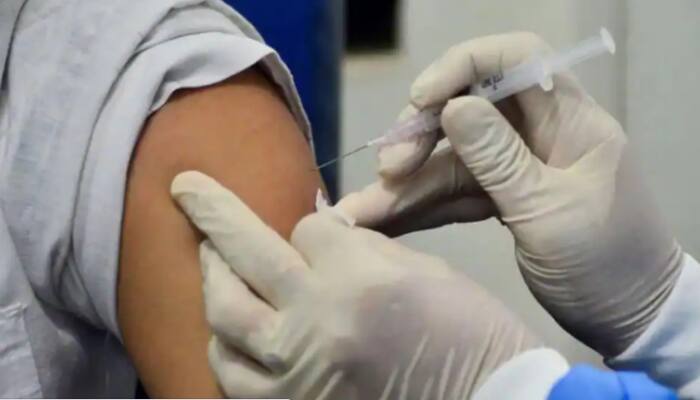
How many tested COVID positive after first and second dose of vaccine in India? Check details here
Out of which 93 lakh received the first dose and out of that 4,208 (0.04 per cent) people got the infection which is four per 10,000 individuals. About 17,37,178 people received the second dose of which only 695 (0.04 per cent) tested positive for COVID-19, ICMR Director General Balram Bhargava said.
Apr 21, 2021, 20:48 PM ISTPeople with THESE health conditions will get COVID-19 vaccine jabs on priority, check full list
In the third phase of the coronavirus vaccination drive which begins from March 1, the government is aiming to expand the vaccination drive to the general public of the nation, which include people with co-morbidities in the 45-59 years age group and senior citizens.
Feb 27, 2021, 22:54 PM ISTFor COVID-19 vaccine in private hospital this is how much you may have to pay
The proposed price range was decided in a meeting between Dr. VK Paul, NITI Aayog and representatives from Serum Institute of India and Bharat Biotech. However, the final call on the price range will be taken by the Union health Ministry.
Feb 27, 2021, 18:05 PM ISTCOVID-19 vaccine price between Rs 300-500, proposes NITI Aayog
The NITI Aayog has promulgated a price range of Rs 300-500 for two Covid vaccines that would be available for the priority group at private facilities in the third phase of vaccination set to begin from March 1, sources privy to the development revealed, adding that the Bharat Biotech’s Covaxin would cost a bit higher.
Feb 27, 2021, 16:51 PM ISTCovaxin only for those above 18 years, not for children: Dr Krishna Ella
Bharat Biotech’s Chairman and Managing Director Dr Krishna Ella interacted exclusively with Zee News.
Jan 14, 2021, 23:38 PM ISTCovaxin trial: AIIMS New Delhi invites volunteers for Phase-III clinical trial
AIIMS Centre for Community Medicine, Dr Sanjay K Rai informed through the advertisement that Phase I/II trials have already been completed.
Dec 24, 2020, 17:02 PM ISTCOVID-19 Vaccine: India’s COVAXIN gets DCGI approval for human clinical trials
The Drug Controller General of India has allowed Bharat Biotech India (BBIL) to conduct human clinical trials for ‘COVAXIN’, a potential COVID-19 vaccine. The human clinical trials will start across India in July 2020. If it emerges successful, COVAXIN will be available by year’s end.
Jun 30, 2020, 21:46 PM IST