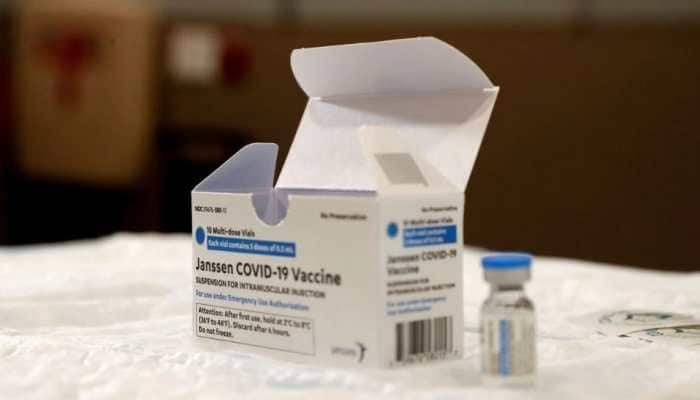
US FDA limits use of Johnson & Johnson Covid-19 vaccine over blood clot risk
US regulators have strictly limited who can receive Johnson & Johnson's COVID-19 vaccine due to the ongoing risk of rare but serious blood clots.
May 06, 2022, 07:20 AM ISTUS approves new booster of COVID-19 vaccines, mix and match dose
“The use of a single booster dose of the Janssen COVID-19 vaccine may be administered at least 2 months after completion of the single-dose primary regimen to individuals 18 years of age and older," FDA said.
Oct 21, 2021, 06:30 AM ISTWHO announces clinical trial to study 3 anti-malarial drugs as potential treatment against COVID-19
The trial involves thousands of researchers in over 600 hospitals in 52 countries.
Aug 13, 2021, 19:17 PM IST'Flushable' pregnancy test gets FDA's approval; set for 2018 launch
Made with a special paper that works long enough to take the test and but then degrades in water, the pregnancy test has no glass and requires no batteries.
Dec 12, 2017, 14:27 PM ISTSilver lining for leukemia patients, new possibility for cure found
The development comes after the US Food and Drug Administration last year approved a cell-based gene therapy, namely the CAR T-cell treatment.
Nov 21, 2017, 18:32 PM ISTSensex little changed; PSU banks down
Moody`s Investors Service upgraded India`s sovereign credit rating for the first time in nearly 14 years on Friday, saying continued progress on economic and institutional reforms would boost the country`s growth potential.
Nov 20, 2017, 12:25 PM ISTIn a first, US regulators approve pill that digitally tracks patients' medication intake
Poor compliance with drug regimens is a common problem in many disease areas, especially when patients suffer from chronic conditions.
Nov 14, 2017, 18:52 PM ISTUS approves first gene therapy for cancer
Gene therapy is a treatment that uses a patient`s own immune cells to fight leukemia.
Aug 30, 2017, 22:50 PM ISTSindoor may carry religious significance, but it is unsafe too! - Read
The FDA’s limit for lead in cosmetics is 20 micrograms per gram. Nineteen percent of the US samples and 43 percent of the India samples exceeded that limit.
Aug 29, 2017, 13:29 PM ISTPersonalised cancer drug approved in US
Pembrolizumab is indicated for the treatment of adult and paediatric patients with unresectable or metastatic solid tumours that have been identified as having a biomarker referred to as microsatellite instability-high (MSI-H) or mismatch repair deficient (dMMR).
May 28, 2017, 18:21 PM ISTFDA clears genetic test to predict Alzheimer's, Parkinson's diseases
The US Food and Drug Administration (FDA) has allowed 23andMe, a genetic testing company, to market its "controversial" direct-to-consumer tests for assessing the risk for 10 diseases or conditions, including Alzheimers and Parkinsons disease.
Apr 08, 2017, 23:59 PM ISTUS FDA approves first drug 'Ocrevus' to treat aggressive multiple sclerosis
US Food and Drug Administration regulators have approved the first drug 'Ocrevus' to treat adult people with an aggressive kind of multiple sclerosis.
Apr 01, 2017, 22:45 PM ISTTexas carries out first US execution of 2017 amid dispute
Texas prison authorities have carried out the first US execution of 2017, as controversy continued to swirl around the drugs used to conduct such lethal injections.
Jan 12, 2017, 10:11 AM ISTLupin receives USFDA nod for hypertension treatment drug in US
Drug major Lupin has received tentative approval from the US health regulator to sell Olmesartan medoximil tablets, used for treating high blood pressure, in the American market.
Dec 26, 2016, 11:46 AM ISTFDA issues warning against homeopathic teething products, says it may harm infants
Consumers should stop using these products and dispose of any in their possession, FDA recommended on Friday.
Oct 01, 2016, 14:38 PM ISTZydus Cadila gets USFDA's final nod for fungal treatment drug
The Zydus Cadila has received final approval from the US Food and Drug Administration (USFDA) to market Voriconazole tablets in strengths of 50 and 200 mg, Cadila Healhtcare said in a BSE filing.
May 25, 2016, 16:19 PM ISTUSFDA actions hurting exports, need govt intervention: Dr Reddy's
Asserting that regulatory action by the US Food and Drug Administration (USFDA) on leading Indian firms has impacted exports from the country, Dr Reddy's Laboratories Chairman Satish Reddy Wednesday asked for a dialogue between government and US health regulator.
Apr 06, 2016, 20:03 PM ISTAurobindo Pharma gets final USFDA nod for anti-viral drug
Aurobindo Pharma has received final approval from the US health regulator to manufacture and market anti-viral Valganciclovir tablets in the American market.
Apr 01, 2016, 13:50 PM ISTBlood pressure drug may treat Alzheimer's disease
A drug used to treat high blood pressure has been found to reduce cell damage often linked to Alzheimer's disease, reports a new study.
Jan 30, 2016, 14:31 PM ISTUS FDA warns of DRL drug import ban if flaws are not fixed
The US Food and Drug Administration (US FDA), which had issued a warning letter to Dr Reddy's Laboratories over quality issues, has said it might withhold approval of the company's fresh drugs and stop import if no corrective action is taken.
Nov 25, 2015, 18:34 PM IST